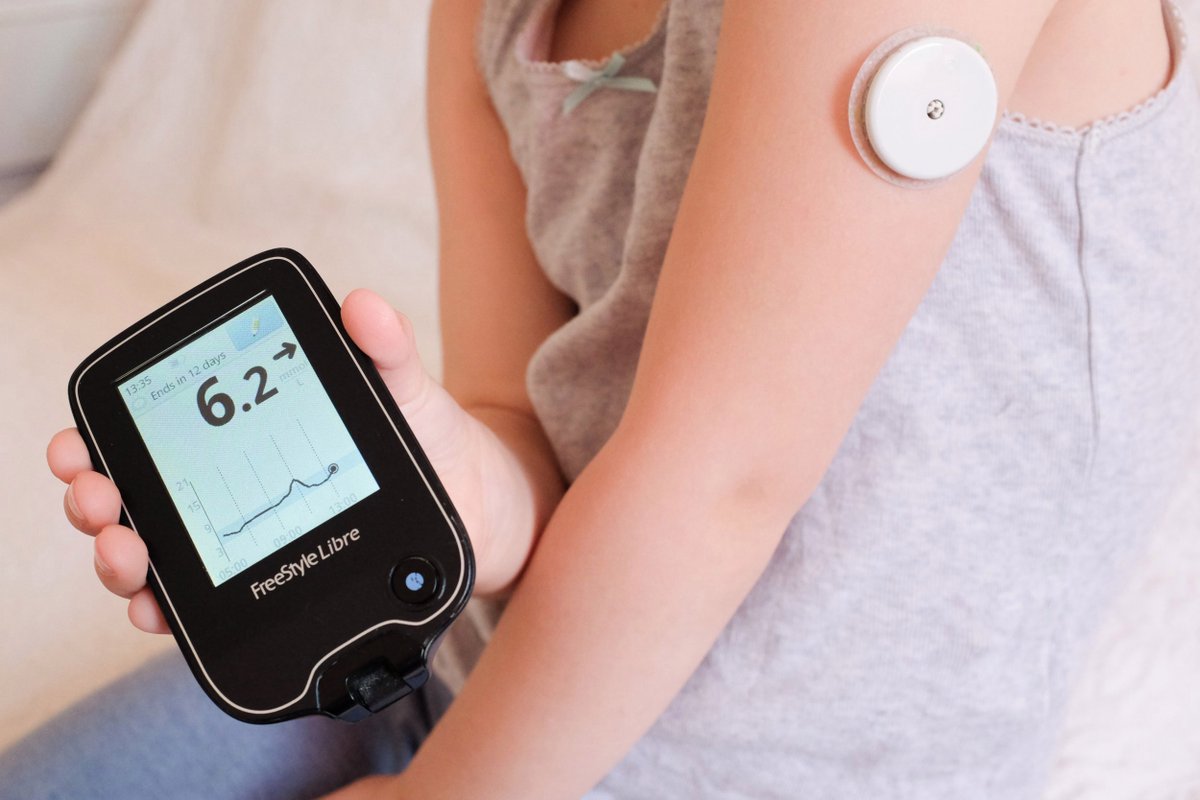 
Nearly 1.5 million Ontarians living with diabetes could benefit from this broad coverage for diabetes support. Alarms further ensure that the patient takes timely action. Libre 2 has the added feature of customized alarms and notifications for low glucose reading, high glucose reading, and loss of signal between the sensor and FreeStyle Libre 2 app or reader. This year, when an upgraded version of Libre 1 (the FreeStyle Libre 2) became available, the government also added the upgrade to the publicly-funded program. The Ontario government first approved the FreeStyle Libre 1 system in September 2019. It is suitable for patients aged 4 and above. FreeStyle Libre 2 is a flash glucose monitor that checks glucose levels with a painless scan instead of fingersticks. Traditional methods for glucose monitoring require a blood sample through a finger prick. Ontario residents who use insulin to manage their Type 1 and Type 2 diabetes can now get the advanced CGM under Ontario’s publicly-funded drug benefit program. It is a Continuous Glucose Monitoring (CGM) system for adults and children with diabetes. The Ontario government has recently announced the coverage for the FreeStyle Libre 2 system. We need governments to offer timely and affordable access to advanced glucose monitoring systems. While newer technologies are being introduced to improve diagnostic and management strategies, this alone is not enough. 
It does not provide real-time alerts or alarms in the absence of a user-initiated action for example, it cannot alert users to low blood glucose levels while they are asleep.Approximately 537 million adults are living with diabetes worldwide this number is projected to rise to 643 million by 2030. Risks associated with use of the system may include hypoglycemia or hyperglycemia in cases where information provided by the device is inaccurate and used to make treatment decisions, as well as mild skin irritations around the insertion site. The FDA evaluated data from a clinical study of individuals aged 18 and older with diabetes, and reviewed the device’s performance by comparing readings obtained by the FreeStyle Libre Glucose Monitoring System to those obtained by an established laboratory method used for analysis of blood glucose. High blood sugar levels can lead to heart disease stroke blindness kidney failure and amputation of toes, feet or legs. When the body doesn’t have enough insulin or cannot use it effectively, sugar builds up in the blood. People with diabetes either do not make enough insulin (type 1 diabetes) or cannot use insulin properly (type 2 diabetes). Typically patients use results of a traditional fingerstick test to make diabetes treatment decisions however, fingerstick testing is not needed to inform appropriate care choices or to calibrate glucose levels with this system.Īccording to the Centers for Disease Control and Prevention, more than 29 million people in the U.S. People with diabetes must regularly test and monitor their blood sugar to make sure it is at an appropriate level, which is often done multiple times per day by taking a fingerstick sample and testing it with a blood glucose meter. “This system allows people with diabetes to avoid the additional step of fingerstick calibration, which can sometimes be painful, but still provides necessary information for treating their diabetes-with a wave of the mobile reader.” Pierre, acting director of the Office of In Vitro Diagnostics and Radiological Health and deputy director of new product evaluation in the FDA’s Center for Devices and Radiological Health. “The FDA is always interested in new technologies that can help make the care of people living with chronic conditions, such as diabetes, easier and more manageable,” said Donald St. It is intended for use in people 18 years of age and older with diabetes after a 12-hour start-up period, it can be worn for up to 10 days. Users can determine glucose levels by waving a dedicated, mobile reader above the sensor wire to determine if glucose levels are too high (hyperglycemia) or too low (hypoglycemia), and how glucose levels are changing. The system reduces the need for fingerstick testing by using a small sensor wire inserted below the skin’s surface that continuously measures and monitors glucose levels. Food and Drug Administration (FDA) approved the FreeStyle Libre Flash Glucose Monitoring System, the first continuous glucose monitoring system that can be used by adult patients to make diabetes treatment decisions without calibration using a blood sample from the fingertip (often referred to as a “fingerstick”).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
